Why aquaculture?
Aquaculture (growing/farming fish for food) is the fastest growing food industry globally. According to the Food and Agriculture Organization (FAO), it has been recording positive annual growth rates since the early 60s (Figure 1). It achieved a new record in 2016 when global fish production through aquaculture reached ~10 kg fish supply per capita, for the first time higher than the supply from capture fisheries (the term for wild fisheries, ie. fish and shellfish captured by fisherman). Today, fish account for 16.6 % of animal protein supply and 6.5 % of total protein for human consumption, and fish has been the major source of animal protein in developing countries of Asia and Africa. Aquaculture is thus an important food-producing sector that contributes high quality protein to support the world’s growing population.

Figure 1. The total amount of wild fish captured by fishing activities (capture fisheries) and the total amount of cultured fish and shellfish produced by aquaculture, in millions of tonnes globally. [Figure from The state of world fisheries and aquaculture, FAO report 2018]
Disease control in aquaculture: an important problem
As in any other animal production industry, diseases are a significant setback for aquaculture. According to a World Bank report from 2014, diseases in aquaculture account for productivity losses of approximately 6 billion USD annually. Since bacteria are major disease agents, excessive administration of antibiotics has been the most commonly applied strategy, especially in countries where legislatory framework on environmental issues is looser.
Taking the case of Vibrio pathogens as an example (eg. V. anguillarum, V. harveyi, V. splendidus, V. alginolyticus, V. parahaemolyticus): they have been named as the “scourge” of marine fish and shellfish. They are the causative agents of a fatal disease known as vibriosis (Figure 2). Vibrios often use live feeds as vehicles to infiltrate the marine hatchery systems, eventually leading to massive mortalities at very early stages of the process (Figure 3).

Figure 2. Massive mortalities caused by vibriosis in different developmental stages. a,b) cultured European seabass, Dicentrarchus labrax, c) cultured European seabass, Dicentrarchus labrax juveniles and d) cultured gilthead sea bream, Sparus aurata larvae in the hatchery. [Figure from Kalatzis et al., 2018]

Figure 3. Facilities for live feed production from a commercial fish farm unit. a) Artemia salina in culture tanks with vigorous aeration, where the naturally present presumptive Vibrio load is regularly estimated between 10^7 and 10^8 cells per mL; b) Brachionus plicatilis culture tanks, where the naturally present presumptive Vibrio load is regularly between 10^2 and 10^8 cells per mL. [Figure from Kalatzis et al., 2018]
Antibiotics in aquaculture lead to AMR risk
Antibiotics that are applied in aquaculture (eg. oxytetracycline, florfenicol, tetracycline, flumequine, oxolinic acid, sulphadiazine/trimethroprim) are not different from those intended for human use. Hence, use of antibiotics in aquaculture raises serious concerns about the development of antimicrobial resistance (AMR), but also about antibiotic dispersal and dissemination among humans. In addition, antibiotics in aquaculture are administered through feed, and since sick fish have decreased appetites, a large fraction of the antibiotics end up in the environment. Here they can remain for years, leading to the disturbance of the natural microbiota in the water column and in the sediment.
Vaccination in aquaculture is challenging
Vaccination can be efficient in disease prevention in aquaculture, however there is a limited number of commercially available vaccines that address only a few of the bacterial diseases affecting fish in aquaculture. Further, vaccines cannot be used efficiently for disease control during the larval and fry (juvenile) stages of cultured fish, since their immune system is still in its infancy, and other cultured animals such as shellfish and crustaceans lack an adaptive immune system.
Phages: a promising avenue for controlling aquaculture pathogens
Phage therapy represents a promising alternative to antibiotics when it comes to bacterial disease control in aquaculture (Figure 4). The aquatic environment is one of the most common sources for isolation of phages against pathogens.
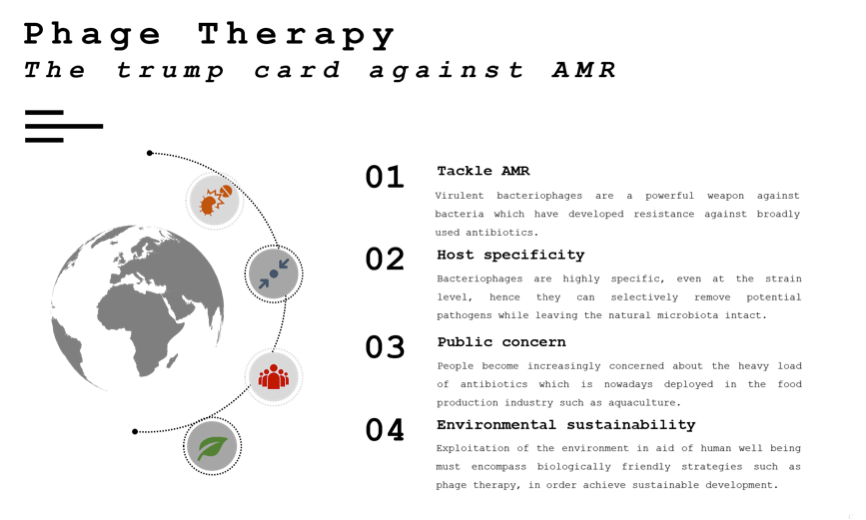
Figure 4. Major advantages of bacteriophage application in the battle against AMR.
The use of phages against pathogenic bacteria in aquaculture was first introduced experimentally in Japan against Lactococcus garvieae in 1999, and it has since been a topic of great interest for the aquaculture-related scientific community. Over the last 20 years, there have been numerous publications describing novel lytic phages that may be applied against bacterial diseases in aquaculture, none of them showing any side effects and all generally increasing survival of the phage-treated cultured organisms.
Phage therapy has been applied in various growth stages of cultured animals such as shrimps, lobsters, sea cucumbers, salmon, turbot, cod with promising results on pathogen control and survival of the cultured organisms. No side effects such as toxicity or stimulation of the immune system (antibody production) have been recorded. The situation in aquaculture is similar whenever phage therapy has been applied against other bacterial pathogens such as species of the genus Flavobacterium, Edwardsiella, Aeromonas, Pseudomonas, Yersinia etc., with numerous publications available in the literature.
How are phages administered?
Injection and immersion are common methods of administration, however, production of phage-coated fish pellets is considered to be the most efficient because it ensures the constant presence of high titer phages in the system via a daily feeding routine. In semi-closed systems like marine hatcheries where the renewal of the water is limited, deployment of antibiotics would disturb the natural microbiota. The reduction of potentially pathogenic bacteria populations that enter such fragile systems could be significantly reduced if phage-based disinfection of live feeds is embraced as part of the daily aquaculture management routine.
It is worth mentioning that the British company Fixed Phage has been quite successful when it comes to phage binding on different substrates, and its products can be customized according to each particular case.
New research into phage control of aquaculture pathogens
A current EU-funded research project, BONUS Flavophage, led by Prof. Mathias Middelboe at the University of Copenhagen explores the possibility of using phage-coated feed to control Flavobacterium pathogens in rainbow trout aquaculture, where there are strong restrictions to the use of antibiotics and increasing concerns about AMR.
After intense sampling at 28 different fish farms (Finland, Sweden, Denmark, Poland, Germany, Latvia and Russia), a collection of 47 phages infecting Flavobacterium psychrophilum, 63 phages infecting Flavobacterium columnare, 119 F. psychrophilum isolates and 133 F. columnare isolates has been established. The objective of the project is to improve the sustainability, food safety, and productivity of aquaculture in the Baltic Sea region by developing environmentally-balanced phage-based strategies to control important Flavobacterium pathogens.
Phage-based products in aquaculture
Very recently, ACD Pharmaceuticals launched its first phage-based product, CuSTuS®, which can be used for effective biocontrol of Yersinia ruckeri; it has so far been quite successful in Norwegian aquaculture. Proteon Pharmaceuticals has launched BAFADOR®, a phage cocktail that claims to target Pseudomonas and Aeromonas infections while stimulating the immune system of fish. Apart from these cases, there are no other phage-based products for aquaculture today in the EU.
Aquatic Biologicals, a new company working toward disease prevention in aquaculture

Aquatic Biologicals is paving the way to sustainable disease prevention and biological control in aquaculture. It is the first marine biotechnology company in Greece and the first EU company that offers a full package of disease surveillance, management, control and prevention in aquaculture.
All aquaculture-related bacterial pathogens are present in the water, so the best way to avoid an outbreak is to keep their populations low. This reduces the likelihood that pathogens will take advantage of the cultured animals and the potential stressors that may occur under aquaculture conditions.
In line with The 2030 Agenda for Sustainable Development, which was shaped by the United Nations, the scope of Aquatic Biologicals encompasses four main sustainable development goals (SDGs):
- SDG 3 (Good Health and Well-being) by developing strategies that limit antibiotic resistance,
- SDG 9 (Industry, Innovation and Infrastructure) by developing production and application strategies for products with commercial potential,
- SDG 12 (Responsible Consumption and Production) by developing non-antibiotic-based disease control measures and
- SDG 14 (Life Below Water) by targeting aquaculture.
Aquatic Biologicals is working toward these goals via three lines of action:
- Development of customized autogenous vaccines against strain-specific diseases.
- Research and development of phage therapy products.
- Advanced surveillance and diagnostic services for the aquaculture industry.
Conclusion
Overall, it is expected that the increasing focus on developing alternatives to antibiotics for disease control in aquaculture, including phage-based products, will reduce the use of antibiotics in this industry and contribute to developing a high-technology sector of sustainable, profitable farming with healthy and high quality animals.
Further reading
Kalatzis, P.G., Castillo, D., Katharios, P. and Middelboe, M. (2018) Bacteriophage Interactions with Marine Pathogenic Vibrios: Implications for Phage Therapy. Antibiotics 7(1). pii: E15. doi: 10.3390/antibiotics7010015.
Kalatzis, P.G., Bastias, R., Kokkari, C., Katharios, P. (2016) Isolation and characterization of two lytic bacteriophages, φSt2 and φGrn1; Phage therapy application for biological control of Vibrio alginolyticus in aquaculture live feeds. PLoS ONE 11(3): e0151101. doi: 10.1371/journal.pone.0151101




